We are
Awamedica
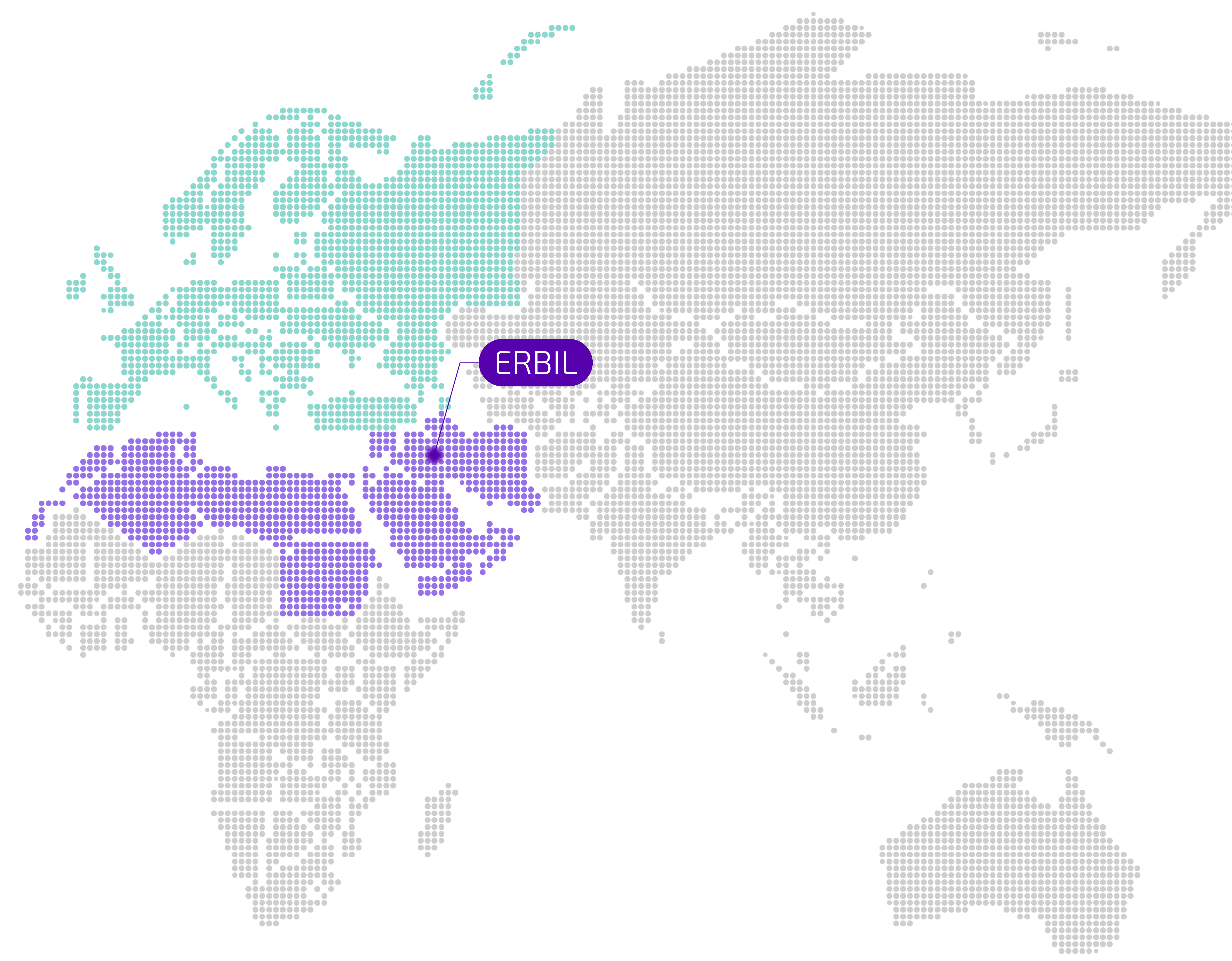
Awamedica is a Class A pharmaceutical manufacturing company headquartered in Erbil, Iraq. Founded in October 2009, our journey is deeply rooted in a commitment to improving healthcare accessibility and quality within Iraq and the wider MENA region. We combine modern manufacturing capabilities with a dedication to scientific rigor, ethical practices, and patient well-being.
At Awamedica, our mission is to improve community health by developing, producing, and marketing a comprehensive portfolio of high-quality, essential pharmaceutical products. We are dedicated to addressing the unmet health needs of the people and ensuring affordable and accessible healthcare for everyone.

Patient-Centric
Every decision is driven by a deep commitment to the health and well-being of each patient we serve.

Quality
Upholding uncompromising standards in all aspects of our operations and products.

Strategic Collaboration
We partner with healthcare professionals and businesses to advance medicine and create mutual growth.
Accessibility
Striving to make essential medicines available and affordable to all who need them.

Community & Sustainability
Building a healthier future through environmentally sustainable practices and meaningful initiatives that support the well-being of our communities.

Integrity & Ethics
We operate with transparency and ethical principles in every action we take.
Our Commitment to Quality
cGMP is the global benchmark for pharmaceutical manufacturing systems, Awamedica adheres strictly to cGMP guidelines in all our operations, guaranteeing the consistency, safety, and quality of every medicine we produce, from raw material sourcing to finished product.

Our commitment to excellence extends to our management systems, certified by internationally recognized ISO standards. We hold ISO 9001 (Quality Management System), ISO 14001 (Environmental Management System), and ISO 45001 (Occupational Health and Safety Management System) certifications.

Awamedica undergoes regular, independent external audits by reputable international bodies. These audits provide an objective assessment of our quality systems, manufacturing processes, and compliance with global standards.

Committed to Excellence
Dedicated to Health.

Tablets
& Capsules
Units

Liquids
Bottles

Semisolid
Tubes

Suspension & Sachet
Dry Powder
Pharmacovigilance, Ensuring Patient Safety
At Awamedica, the well-being of every patient is paramount. Our robust Pharmacovigilance system is dedicated to continuously monitoring the safety and performance of our medicines once they reach the market.
Pharmacovigilance Info


